Dr. Nisar Ullah

Professor, Graduate Studies Coordinator
Our research interests are broadly in the area of synthetic organic chemistry, with a particular focus on the synthesis of bioactive natural products and novel molecular architectures.
📍 Bldg. 4-218
☎ (+966) 13 860 7527
🔗 ORCID: 0000-0002-8172-2771
🔗 Scopus: 8154097400
Research Area
⬡ Synthetic Organic & Medicinal Chemistry
Welcome to the Synthetic Organic Chemistry group
Organic synthesis is a cornerstone of chemistry, and our research is centered on this discipline. We focus on the design and synthesis of organic molecules with meaningful functions, including biological activity such as natural products, pharmaceuticals, and drug-like compounds as well as materials with valuable properties. Our goal is to tackle complex challenges that require a deep understanding of fundamental principles to access intricate molecular architectures, whether derived from natural sources or developed through rational design.
Our research interests include:
Total Synthesis:
Total synthesis refers to the complete chemical construction of organic molecules from simple, commercially available, or naturally occurring starting materials. Our work in this area emphasizes the development of innovative synthetic strategies and the careful selection of organic transformations to efficiently build structurally complex, biologically active natural products.
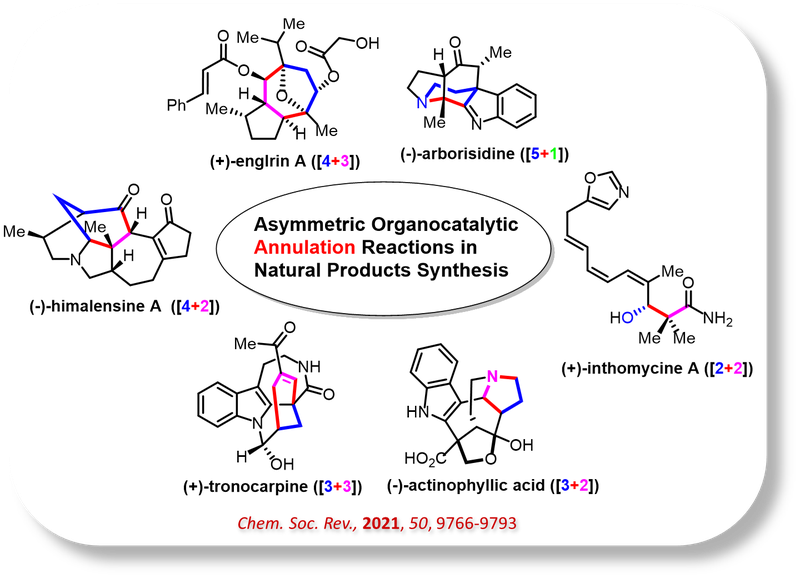
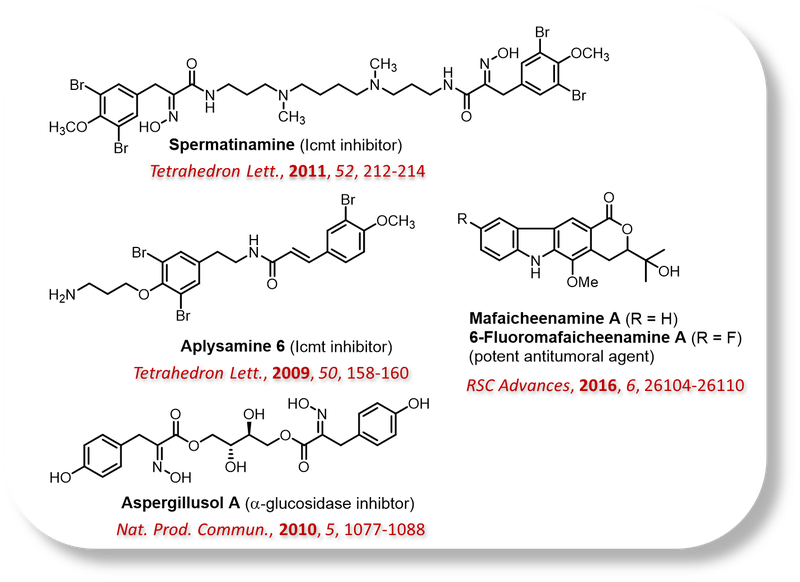
New Methodologies in Organic Synthesis:
Our research in this area focuses on developing innovative tools and strategies for efficient synthetic transformations aimed at constructing novel molecular architectures. This involves the discovery and advancement of new reactions through the design of reagents and catalysts. The outcomes of these efforts are closely aligned with and contribute directly to our work in total synthesis and medicinal chemistry.
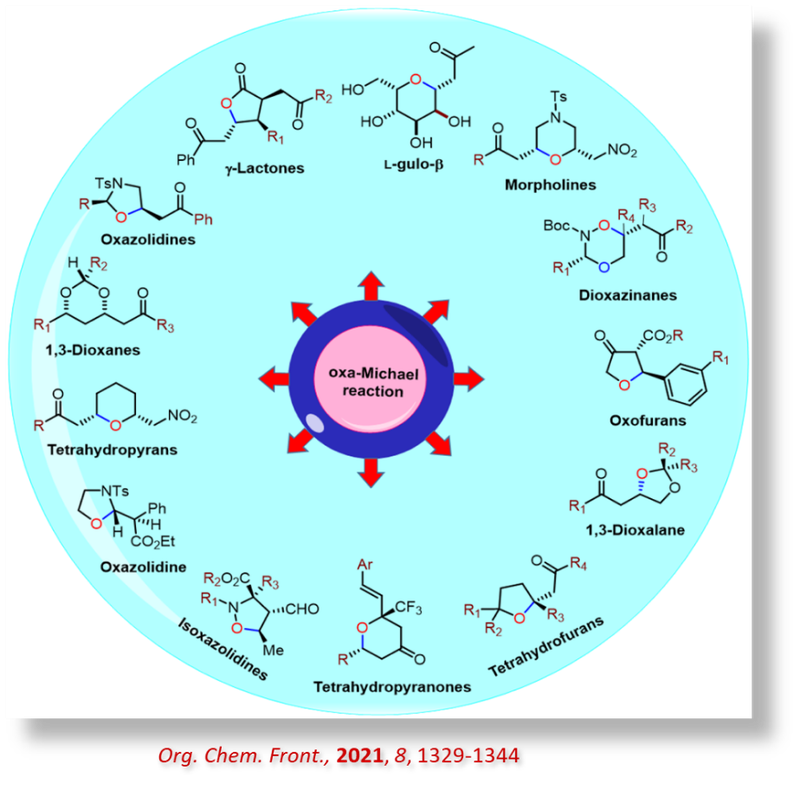
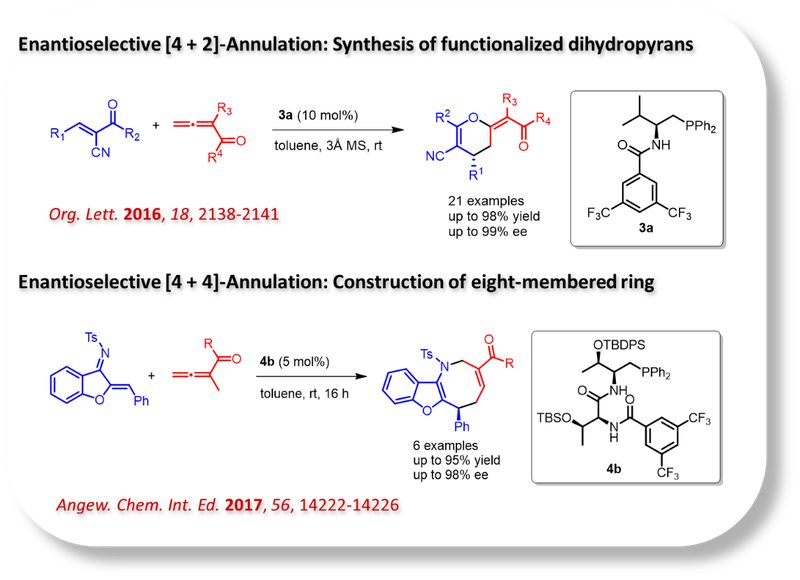
Medicinal Chemistry:
This research area focuses on the optimization of lead compounds and the synthesis of structural analogues of bioactive motifs. Improvements in biological activity are achieved through targeted modifications of the molecular structure of the lead compound, enabling the development of more effective and selective candidates.
Education
✔︎ PhD: University of Graz, 2001
✔︎ Postdoc: McMaster University, 2002
✔︎ Postdoc: University of Guelph, 2003
✔︎ BSc: University of Peshawar, 1993
- M. Ashraf, M. L. Firmansyah, S. Abdel-Azeim, N. Ullah*. The design of β-carboline–8-aminoquinoline tethered probes for nanomolar Zn²⁺ sensing and cell imaging. Sens. Actuator B-Chem., 2026, 455, 139645.
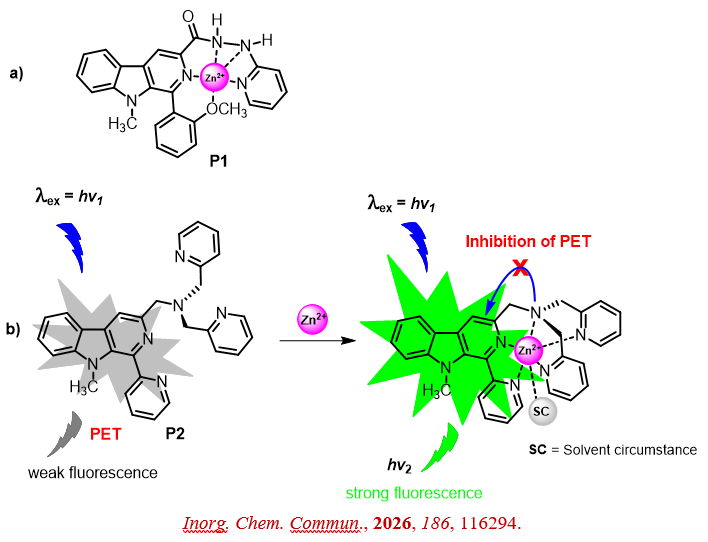
- M. Ashraf, M. L. Firmansyah, S. Abdel-Azeim, N. Ullah*. Biocompatible β-carboline–derived fluorescent probes for nanomolar detection and intracellular Zn2+ imaging. Inorg. Chem. Commun., 2026, 186, 116294.
- T. N. Jahangir, M. Ashraf, S. Abdel-Azeim, S. Khan, N. Ullah, T. A. Kandiel. Cobalt-based molecular catalysts designed for significantly enhanced low-bias photoelectrochemical water oxidation over BiVO4. ACS Catal., 2026, 16, 356−369.
- M. Ashraf, M. L. Firmansyah, S. Abdel-Azeim, R. J. K. Susilo, S. Hayazad, N. Ullah*. β-Carboline platform-based novel fluorescent probes for the selective detection of zinc ions: synthesis, live-cell imaging and computational studies. Analyst, 2025, 150, 5223
- M. Al-Sharafi, M. Ashraf, M. L. Firmansyah, S. Chigurupati, El H. Anouar, N. Ullah*. Design and synthesis of β-carboline hydrazone hybrids as new α-amylase and α-glucosidase inhibitors: Inhibition kinetics and molecular docking studies. Asian J. Org. Chem., 2025, 14, e00365 (Invited Article).
- M. L. Firmansyah, G. A. N. Rohman, N. Ullah*. Machine-Learning-Optimized Palm-Biomass-Derived Activated Carbon Adsorbent for Gold Recovery from Mobile Leachate. ACS Sustainable Resour. Manage., 2025, 2, 825–832.
- T. Ahmad, S. Abdel-Azeim, S. Khan, N. Ullah*. Dinitrophenyl and NBD platforms based fluorescent sensors for nanomolar detection of zinc ions: Synthesis, zinc ions sensing and DFT studies. Inorg. Chem. Commun., 2024, 170, 113151.
- M. Ashraf, N. Ullah, I. Khan, W. Tremel, S. Ahmad, M. N. Tahir. Photoreforming of waste polymers for sustainable hydrogen fuel and chemicals feedstock: Waste to energy. Chem. Rev., 2023, 123, 4443–4509.
- M. Ashraf, M. S. Ahmad, Y. Inomata, N. Ullah*, M. N. Tahir, T. Kida. Transition metal nanoparticles as nanocatalysts for Suzuki, Heck and Sonogashira cross-coupling reactions. Coord. Chem. Rev., 2023, 473, 214928.
- C. X. A. Tan, R. Li, F. Zhang, L. Dai, N. Ullah*, Yixin Lu*. Synthesis of axially chiral CF3-substituted 2-arylpyrroles by sequential phosphine-catalyzed asymmetric [3+2] annulation and oxidative central-to-axial chirality transfer. Angew. Chem. Int., Ed., 2022, 61(47), e202209494.
- T. Ahmad, S. Khan, N. Ullah*. Recent advances in the catalytic asymmetric Friedel-Crafts reactions of indoles. ACS Omega, 2022, 7, 35446–35485 (Invited Review Article).

Dr. Mochamad Lutfi Firmansyah
Postdoc Fellow

Dr. Ilma Amalina Suyanto
Postdoc Fellow

Muhammad Dani Sirait
PhD Student

Salman Saeed Sheikh
PhD Student

Nayab Mohsin
PhD Student

Samba Joof
MS Student

Yousef Alalwan
MS Student
| Undergraduate Courses Taught |
|---|
|
• CHEM 101: General Chemistry I (Lectures, and Laboratory). • CHEM 201: Organic Chemistry I (Lectures, and Laboratory). • CHEM 204: Organic Chemistry II • CHEM 202: Organic Chemistry II LAB • CHEM 305: Organic Synthesis and Characterization Laboratory • CHEM 403: Synthetic Organic Chemistry • CHEM 406: Spectroscopic Identification of Organic Compounds |
| Graduate Courses Taught |
|---|
|
• CHEM 503: Organic Reactions: Mechanism and Reactivity • CHEM 536: Spectroscopic Identification of Organic Compounds • CHEM 532: Synthetic Organic Chemistry |